Hydrogen
Isotopes, Preparation, Properties and Uses of hydrogen
Preparation (H2O2) : is first prepared by Jhenard
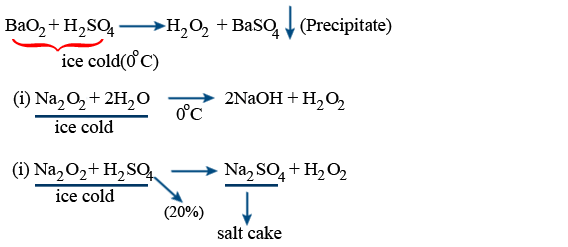

Barium sulphate taken in dry (or) powder form will form BaSO4 at the top and stops the reaction (or) formation of H2O2. So BaO2 is taken as paste

Merck Process :
BaO_{2} + CO_{2} + H_{2}O \rightarrow H_{2}O_{2} + BaCO_{3}\downarrow
Industrial Process :
It is electrolytic oxidation of 50% ice cold H2SO4
Cathode : Lead
Anode : Pt
electrolyte : 50% H2SO4
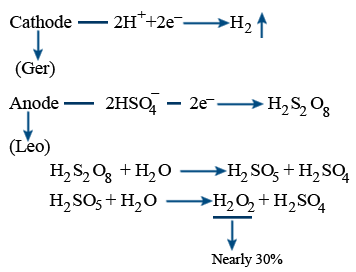
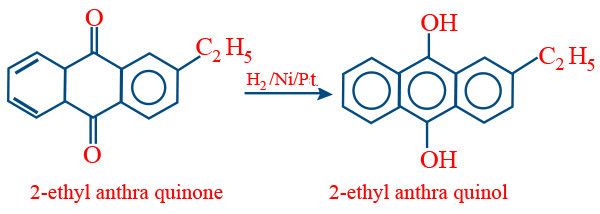
Auto oxidation (or) Arial oxidation :
It is a cyclic process
Part1: View the Topic in this Video from 0:05 to 5:58
Part2: View the Topic in this Video from 0:06 to 9:17
Part3: View the Topic in this Video from 0:05 to 11:46
Disclaimer: Compete.etutor.co may from time to time provide links to third party Internet sites under their respective fair use policy and it may from time to time provide materials from such third parties on this website. These third party sites and any third party materials are provided for viewers convenience and for non-commercial educational purpose only. Compete does not operate or control in any respect any information, products or services available on these third party sites. Compete.etutor.co makes no representations whatsoever concerning the content of these sites and the fact that compete.etutor.co has provided a link to such sites is NOT an endorsement, authorization, sponsorship, or affiliation by compete.etutor.co with respect to such sites, its services, the products displayed, its owners, or its providers.
1. Degree of hardness (in ppm) = \tt \frac{wt. \ of \ CaCO_{3}\left(g\right)}{wt. \ of \ hard \ water \ \left(g\right)}\times 10^{6}

