Haloalkanes and Haloarenes
Polyhalogen Compounds
- DICHLORO ALKANES:
Preparations: 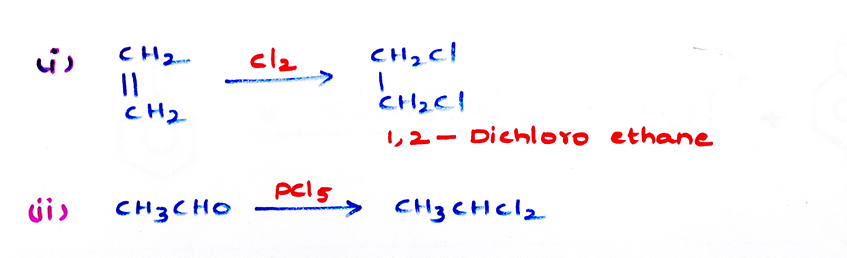
(iii) Dichloro methane is prepared by partial reduction of CHCl3
(iv) 1° iodo compounds are unstable. They decompose to give alkene and I2.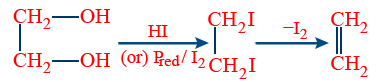
- Properties of dichloro alkane:
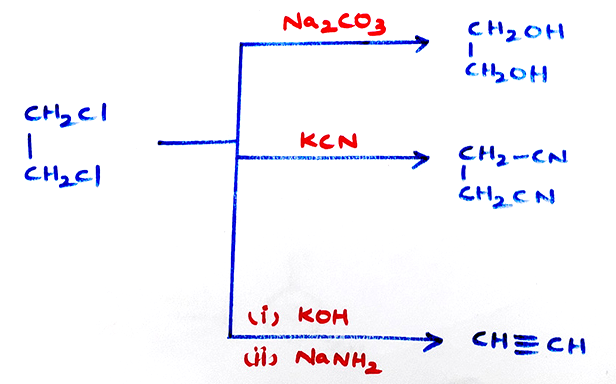
- (*) CH2Cl2 is colourless, highly poisonous sweet smelling liquid. It is used as medium in ozonolysis reactions.
- D.D.T:
- 2, 2 Dis (P-chlorophenyl) - 1,1,1, Trichloroethane.
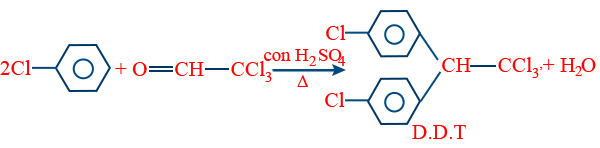
(b) It is an insecticide but harmful to animals and birds for their fertility.
(c) The artificial sweetener sucralose (splenda) is trichloro derivative of sucrose and is also named as DDT lite.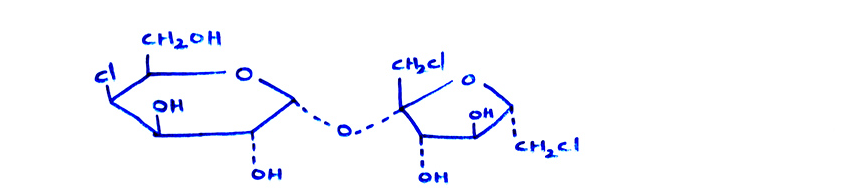
Sucralose (or) splenda (or) DDT lite- Freons, Chloro fluoro carbons:
(i) CF3Cl (Freon - 13), CF2Cl2 (Freon - 12), CFCl3(Freon - 11) etc., At one time, freon - 12 was most commonly used as refrigerant and propellant in aerosols.
(ii) \tt 3CCl_4+2SbF_3\xrightarrow[]{{SbCl_5/ \Delta}}3CF_2Cl_2+2SbCl_3
(iii) \tt CCl_4+2HF\xrightarrow[]{{SbCl_3/ \Delta}}CF_2Cl_2+2HCl
(iv) Its use is being discarded because of its main role in depletion of ozone layer around the earth.
(v) In aerosols, it has been replaced by gaseous butane and isobutane. - Carbon tetrachloride (CCl4):
(a) \tt CH_4+4Cl_2 (large \ excess)\xrightarrow[]{{h\nu}}CCl_4+4HCl
(b) \tt CS_2+3Cl_2 \xrightarrow[]{{AlCl_3/30^{o}C}}CCl_4+S_2Cl_2
(c) CS2 + 2S2Cl2 → CCl4 + 6S
(d) It is used as solvent for oils, fats, etc., as fire extinguisher for electrical fires under the name pyrene fire extinguisher as a medicine for hook worms, etc., - Properties
(a) \tt CCl_4+H_2 \xrightarrow[]{{Fe/Steam}}CHCl_3+HCl
(b) \tt CCl_4\xrightarrow[-KCl]{{4KOH}}C(OH)_4\xrightarrow[-H_2O]{{}}CO_{2}\xrightarrow[]{{2KOH}}K_{2}CO_{3}+H_{2}O
(c) \tt 3CCl_4+2SbF_3\xrightarrow[]{{SbCl_5}}3CF_{2}Cl_2+2SbCl_{3}
(d) CCl4 causes liver cancer and depletes O3 layer.
(e) Vapours of CCl4 get oxidised by steam to give poisonous phogene (gas)
CCl4 + H2O → COCl2 + 2HCl - Haloforms: (CHX3)
(i) CHCl3 chloroform is the main chemical of this group. It is used as solvent for organic compounds and for the preparation of many other organic chemicals.
(ii) Long back it was used as anesthetic.
(iii) Long time exposure to CHCl3 may cause damage to liver and kidney. - Preparations:
(a) Methyl ketones eg. acetone and methyl carbinols CH3CH(OH)-R, on heating with paste of bleaching powder (or) Cl2/dil NaOH produce CHCl3.
(b) CaOCl2 + H2O → Ca(OH)2 + Cl2
\tt CH_{3}-COCH_{3}\xrightarrow[]{{Cl_2}}CCl_3 COCH_3\xrightarrow[]{{Ca(OH)_2}}CHCl_3+(CH_3COO)_2 Ca
CH3COCH3 + 6Cl2 + Ca(OH)2 → 2 CHCl3 + (CH3COO)2Ca + 6 HCl
(c) \tt CH_{3}COCH_{3}+3Cl_2+4 NaOH\xrightarrow[]{\Delta}CHCl_3 + CH_3 COONa+3NaCl+3H_2 O
(d) \tt CH_{3}CH(OH)-R+4Cl_2+6 NaOH\xrightarrow[]{\Delta}CHCl_3 + RCOONa+5NaCl+5H_2 O
(e) Pure chloroform is prepared from chloral hydrate.
\tt CCl_{3}CHO.H_2O+NaOH\xrightarrow[]{\Delta}CHCl_3 + HCOONa+H_2 O
(g) Concentrated NaOH, if used in the above reaction, Cannizzaro reaction will take place. CHCl3 will be a minor product
CCl_{3}CHO\xrightarrow[]{NaOH(Conc)/ \Delta}CCl_3 CH_2OH+CCl_3 COONa
(h) CCl_{4}+2[H]\xrightarrow[]{Fe/Steam}CHCl_3 +HCl
(i) Preparation of iodoform (Yellow solid) is used as a test of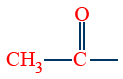 and CH3CH(OH)-groups in compounds, commonly called as iodoform test.
and CH3CH(OH)-groups in compounds, commonly called as iodoform test.
(k) CHI3 is yellow solid and used as an antiseptic. It is prepared in similar manner as CHCl3 but I2 solution is taken in place of Cl2 water. Industrially, CHI3 is prepared by the electrolysis of a solution containing KI, Na2CO3 and C2H5OH
\tt KI\rightleftharpoons K^{+} + I^{-}
At anode 2I− → l2 + 2e−
C2H5OH + 4l2 + 3Na2CO3 → CHl3 + HCOONa + 5NaI + 3CO2 + 2H2O. - Properties:
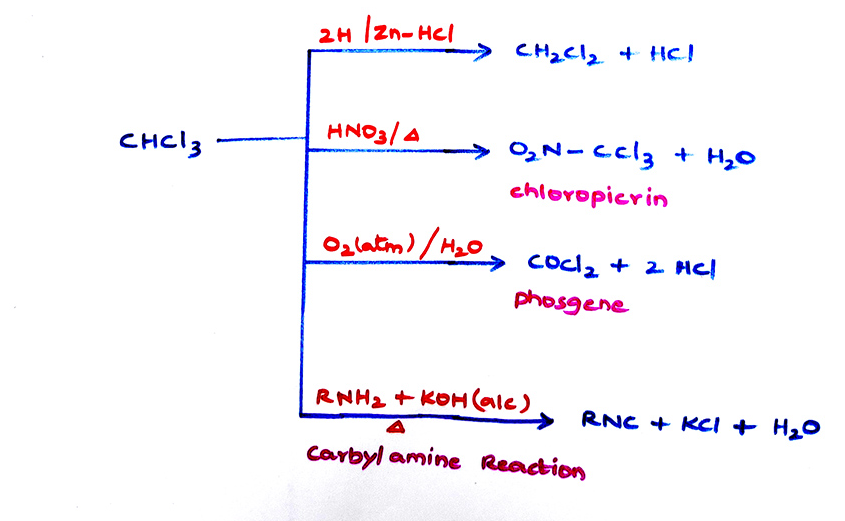
- Tear gas O2N - CCl3 can also be prepared as
\tt CH_{3}NO_{2}\xrightarrow[]{{Cl_{2}/dil\ NaOH}}CCl_{3}NO_{2}
0.6 to 1.0% Et-OH of the quantity of CHCl3 is added to slow down the aerial oxidation and remove any phosgene formed.
COCl2 + 2C2H5OH → (C2H5)2CO3 + 2HCl. - (*) CnF2n+2 are called perflurocarbons.
- (*) \tt C_{7}H_{16}(vapours)+16F_{2}\xrightarrow[]{{H\nu}}C_{7}F_{16}(perfluroheptane)+16HF
View the Topic in this Video from 38:25 to 53:20
Disclaimer: Compete.etutor.co may from time to time provide links to third party Internet sites under their respective fair use policy and it may from time to time provide materials from such third parties on this website. These third party sites and any third party materials are provided for viewers convenience and for non-commercial educational purpose only. Compete does not operate or control in any respect any information, products or services available on these third party sites. Compete.etutor.co makes no representations whatsoever concerning the content of these sites and the fact that compete.etutor.co has provided a link to such sites is NOT an endorsement, authorization, sponsorship, or affiliation by compete.etutor.co with respect to such sites, its services, the products displayed, its owners, or its providers.

