Co-ordination Compounds
Nomenclature and Isomerism
| General Formula | No. of Geometrical Isomers | No. of Optically Active Isomers | No. of Inactive Isomers | Total stereo |
| Ma6 | 1 | 0 | 1 | 1 |
| Ma5b | 1 | 0 | 1 | 1 |
| Ma4b2 | 2 | 0 | 0 | 2 |
| Ma4bc | 2 | 0 | 0 | 2 |
| Ma3b3 | 2(1 facial, 1 meridian) | 0 | 0 | 2 |
| Ma3b2c | 3(1C+2T) | 0 | 3 | 3 |
| Ma3bcd | 4(1C+3T) | 2 | 3 | 5 |
| Ma2b2c2 | 5(1C+4T) | 2 | 4 | 6 |
| Ma2b2cd | 6(2C+4T) | 4 | 4 | 8 |
| Ma2bcde | 9(6C+3T) | 12 | 3 | 15 |
Prefect complexes:
(i) Stable compounds
(ii) Will not give the test of constituent ions.
Imperfect complex:
(i) Relatively less stable compounds
(ii) Will give the test of the constituent ions.
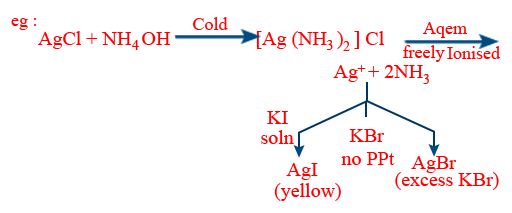
∴[Ag(NH3)2]+ is a perfect complex towards KBr
imperfect complex towards excess KBr and KI.
Nomenclature of Co ordination compounds:
IUPAC Nomenclature:
1. The central atom is listed first.
2. The ligands are then listed in alphabetical order. The placement of a ligand in the list does not depend on its charge.
3. Polydentate ligands are also listed alphabetically. The first letter of the abbreviated ligand is used to determine the alphabetical order.
4. No space should be left between and the metal within a coordination sphere.
5. The charge of a charged complex cation or anion without the counter ion, is to be written outside the square brackets as a right subscript.
Ex: [Cr(H2O)6]3+
Part1: View the Topic in this Video from 12:27 to 58:16
Part2: View the Topic in this Video from 0:40 to 47:42
Disclaimer: Compete.etutor.co may from time to time provide links to third party Internet sites under their respective fair use policy and it may from time to time provide materials from such third parties on this website. These third party sites and any third party materials are provided for viewers convenience and for non-commercial educational purpose only. Compete does not operate or control in any respect any information, products or services available on these third party sites. Compete.etutor.co makes no representations whatsoever concerning the content of these sites and the fact that compete.etutor.co has provided a link to such sites is NOT an endorsement, authorization, sponsorship, or affiliation by compete.etutor.co with respect to such sites, its services, the products displayed, its owners, or its providers.

