Aldehydes, Ketones and Carboxylic Acids
Preparation of Aldehydes and Ketones
Preparations :
(A) From Hydrocarbons :
(a) Wacker process :

(b) Ozonolysis :
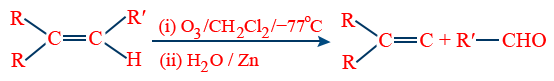
c) Hydroboration Oxidation of alkyne :
(i) Aldehyde can not be prepared by using B2H6 / THF from terminal C ≡ C. For this purpose disiamyl borane is used.
d) From toluene :
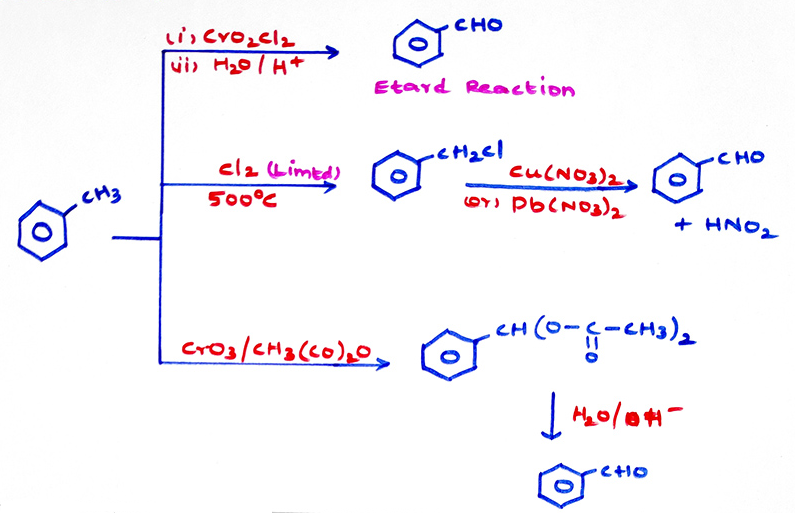
1) Friedel - crafts Acylation does not take place in nitro benzene benzaldehyde and benzoic acid, in which the benzene ring has been highly deactivated.
2) Grignard reagent must be used in limited quantity, other wise the ketone formed will finally give 3° alcohol, the ketone has to be distilled out side by side
View the Topic in this Video from 3:45 to 43:35
Disclaimer: Compete.etutor.co may from time to time provide links to third party Internet sites under their respective fair use policy and it may from time to time provide materials from such third parties on this website. These third party sites and any third party materials are provided for viewers convenience and for non-commercial educational purpose only. Compete does not operate or control in any respect any information, products or services available on these third party sites. Compete.etutor.co makes no representations whatsoever concerning the content of these sites and the fact that compete.etutor.co has provided a link to such sites is NOT an endorsement, authorization, sponsorship, or affiliation by compete.etutor.co with respect to such sites, its services, the products displayed, its owners, or its providers.

