Environmental Chemistry
Atmospheric Pollution by Particulate Pollutants
Smog: (Smoke + Fog)
1. Classical 2. Photo chemical
(i) Classical (Reducing / London)
eg: humid, cool climate reducing mixtures, smoke + SO2 + Fog
(ii) Photo chemical: (Oxidising / LA)
→ Fossil fuels (unburnt)
→ Hot, dry climate
→ Oxidising mixture
2NO + O2 → 2NO2
NO2 + O3 → NO2 + O2 + O
O2 (air) + O → O3 (toxic)
O3 + unburnt HC → HCHO
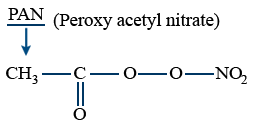
O3 + PAN → eye irritant
O3 + NO → nose, throat
Corrosive action on rubber, paint, buildings...
Some plants can metabolise nitrogen oxide.
Part1: View the Topic in this Video from 0:08 to 15:21
Part2: View the Topic in this Video from 0:08 to 10:48
Disclaimer: Compete.etutor.co may from time to time provide links to third party Internet sites under their respective fair use policy and it may from time to time provide materials from such third parties on this website. These third party sites and any third party materials are provided for viewers convenience and for non-commercial educational purpose only. Compete does not operate or control in any respect any information, products or services available on these third party sites. Compete.etutor.co makes no representations whatsoever concerning the content of these sites and the fact that compete.etutor.co has provided a link to such sites is NOT an endorsement, authorization, sponsorship, or affiliation by compete.etutor.co with respect to such sites, its services, the products displayed, its owners, or its providers.

